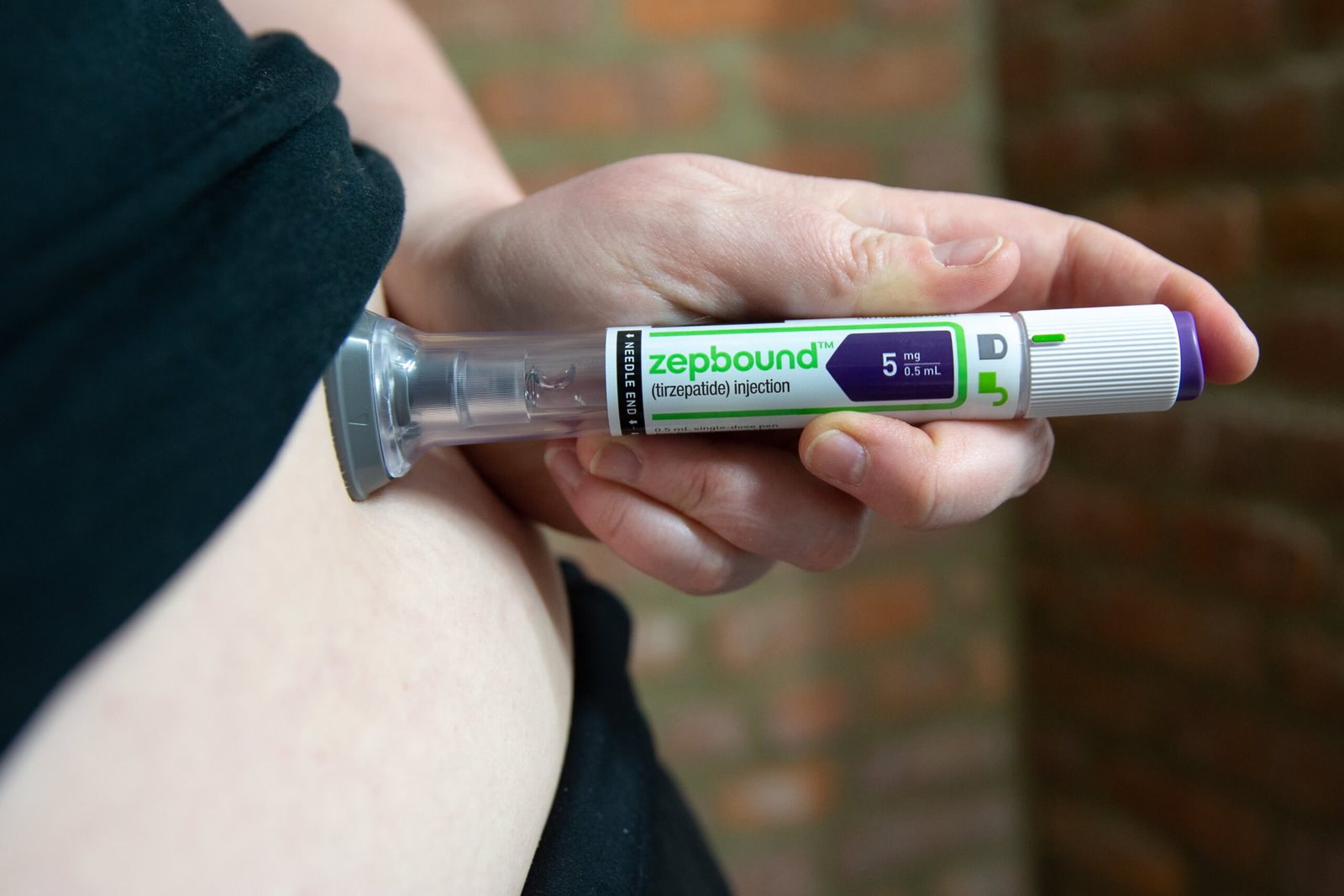
In a major step forward for obesity treatment, Eli Lilly introduces a new version of its obesity drug Zepbound, offering a month’s supply of doses in a single pen. The announcement marks another milestone for the pharmaceutical giant as it continues to expand access and convenience for patients navigating their weight management journey.
On Monday, Eli Lilly unveiled a new multi-dose format of its highly popular obesity medication, Zepbound. The updated version allows patients to receive four weekly doses from a single device, simplifying the treatment process significantly.
What’s New: The KwikPen Rollout
The newly introduced device, known as the KwikPen, is now available for cash-paying patients through Lilly’s direct-to-consumer platform, LillyDirect. Pricing begins at $299 per month for the lowest dosage tier, making it a competitive option for eligible patients paying out of pocket.
Unlike the previous format — where patients had to use a separate single-dose auto-injector each week — the KwikPen combines four weekly injections into one convenient device. This reduces the hassle of managing multiple pens and offers a smoother experience overall.
Previously, patients were required to use either:
-
A single-dose auto-injector device each week, or
-
Single-dose vials that required drawing medication into a syringe before injection
The multi-dose pen eliminates much of this complexity, making adherence easier and more practical.
Why This Move Matters
Since its launch in late 2023, Zepbound has experienced extraordinary demand, quickly becoming one of the most talked-about medications in the weight loss space. Lilly’s direct-to-consumer strategy has played a crucial role in fueling this growth. By introducing a more user-friendly format on LillyDirect, the company is positioning itself to attract even more patients seeking accessible treatment options.
The impact of Zepbound on the market has been substantial. In the fourth quarter alone, the drug generated $4.2 billion in U.S. revenue — representing a staggering 122% year-over-year increase. This surge helped Eli Lilly capture a dominant share of the rapidly expanding weight-loss drug market, outpacing rival Novo Nordisk.
Regulatory Backing and Expanded Approval
The company also confirmed that the U.S. Food and Drug Administration approved a label expansion for Zepbound to include the new multi-dose device. This regulatory clearance reinforces confidence in the updated delivery system and ensures it meets safety and efficacy standards.
Importantly, the KwikPen itself is not new technology. It has already been used successfully for other medications, including Lilly’s widely prescribed diabetes treatment, Mounjaro. Its familiarity among patients and healthcare providers may further accelerate adoption.
Company Perspective
Ilya Yuffa, President of Lilly USA and Global Customer Capabilities, emphasized that the move aligns with the company’s broader mission to support individuals living with obesity.
He noted that introducing the Zepbound KwikPen offers patients a device already trusted worldwide for other Lilly medicines, while making treatment more convenient and accessible.
The Bigger Picture
The introduction of a multi-dose device represents more than just a packaging update — it reflects how pharmaceutical companies are evolving to meet patient preferences. Convenience, affordability, and streamlined access are increasingly shaping the competitive landscape in the obesity drug market.
With demand for weight management therapies continuing to surge, Eli Lilly introduces a new version of its obesity drug Zepbound, offering a month’s supply of doses in a single pen — reinforcing its commitment to innovation, accessibility, and long-term leadership in this fast-growing space.